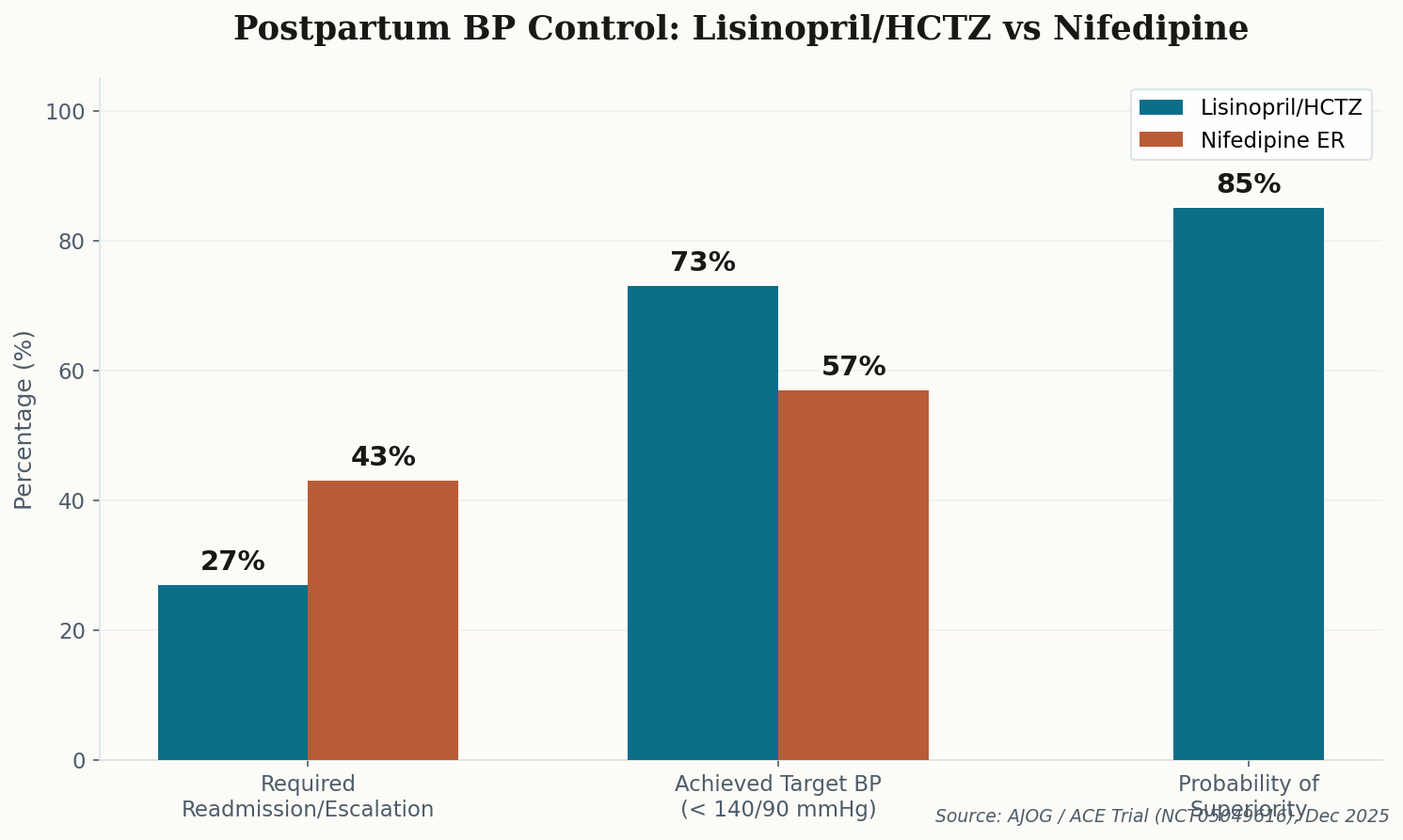
The $745 Bottle That Used to Cost $15

Here's a number that should make your blood boil: $745. That's what a single bottle of Qbrelis—the only FDA-approved liquid form of Lisinopril—costs right now in early 2026. The same medication in tablet form? Four dollars a month. Generic. Everywhere. No drama.
The liquid version isn't a luxury. It's a necessity for children with congenital hypertension and elderly patients who can't swallow pills. And right now, a severe supply shortage has made it nearly impossible to find. Clinicians are being forced into workarounds: compounded suspensions from specialty pharmacies (inconsistent quality, insurance won't cover them), or pivoting patients to Enalapril solution—a different drug entirely.
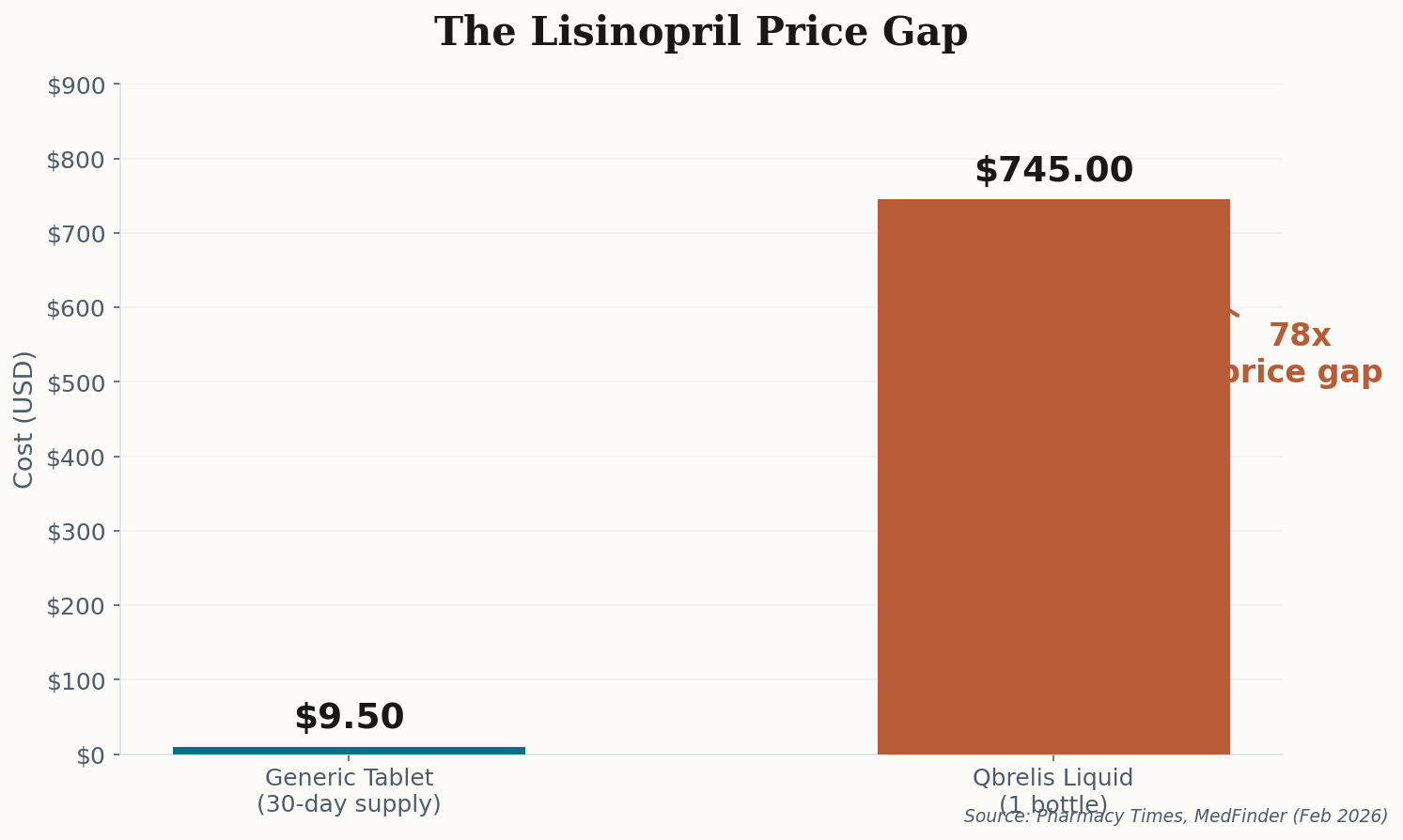
This is the pharmaceutical access paradox in miniature. The active ingredient is dirt cheap—among the most affordable generics on Earth. But the moment you need it in a different delivery form, you're at the mercy of a single manufacturer and a supply chain that apparently can't handle one factory hiccup without leaving the most vulnerable patients scrambling.
What to watch: If you or a family member takes liquid Lisinopril, talk to your pharmacist about compounded alternatives now—don't wait for the shortage to resolve. Ask your prescriber whether an Enalapril solution switch is clinically appropriate.